LEGISLATIVE UPDATES
State and federal laws and regulations allow PDMPs the authority to operate, detail the authorized users, and establish criteria for the querying and use of the PDMP data. TTAC provides comprehensive quarterly and annual summaries of proposed and enacted laws and regulations affecting PDMPs.
PROPOSED AND ENACTED PDMP LEGISLATION AND REGULATION QUARTERLY REPORT
(Q1 CY 2020 THROUGH Q4 CY 2024)
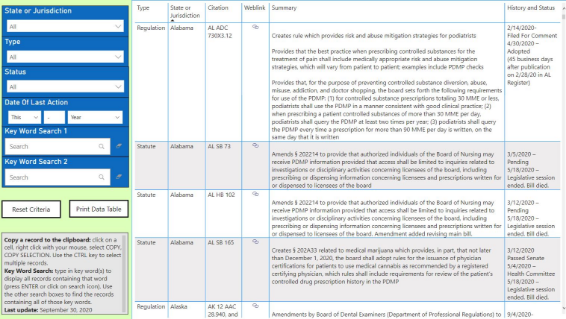
This tool is intended to inform interested stakeholders, including PDMP administrators and state legislators, about statutes and regulations introduced at the state and federal levels related to PDMPs. TTTAC will also track their progress throughout the year with quarterly updates. The full language is available by clicking on the weblink next to the citation reference.
TTAC VisualizationCOMPILATION OF PDMP STATUTES AND REGULATIONS BY TOPIC THROUGH Q4 CY 2024
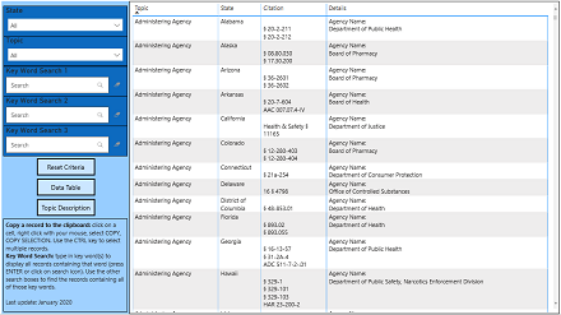
In order to provide an in-depth overview of prescription drug monitoring programs (PDMPs) across the United States, the PDMP Training and Technical Assistance Center (PDMP TTAC) located at the Institute for Intergovernmental Research (IIR) has compiled a complete listing of the statutes and regulations, broken down by topic area, in each state, territory, and jurisdiction with a PDMP.
The table of contents on the following page includes a link to the section related to the named topic. Each section contains a short description of the topic area and tables for each topic listing the statutes and regulations related to that topic for each PDMP as well as summaries of the statutory and/or regulatory requirements in each jurisdiction.
Please note that the information in this document is based on the statutes and regulations in each jurisdiction. Every effort has been made by PDMP TTAC to ensure its accuracy and it will be updated from time to time. Where the information in this table differs from information reported to PDMP TTAC by a state PDMP administrator, the difference is usually due to a state interpreting their law to allow a practice that is not set out specifically in law or rule. Questions regarding the laws and regulations of a specific state should be referred to the PDMP administrator for that state.
TTAC VisualizationResources
Summary of selected bills and regulations enacted or adopted in 2023.
Summary of bills and regulations enacted or adopted in 2022
The document informs PDMP administrators, state and federal legislators, and other interested stakeholders about state and federal legislation of each state and federal agencies related to PDMPs. The document identifies bills and regulations introduced at the state and federal levels related to PDMPs.
21 Code of Federal Regulations (CFR) Parts 1300, 1301, and 1304: Registration Requirements for Narcotic Treatment Programs
View ReportReport — 09/21/2021
In its continuing effort to keep prescription drug monitoring programs (PDMPs) and other stakeholders apprised of new and changing practices, current research, and new laws and regulations, the Prescription Drug Monitoring Program Technical Training and Assistance Center (PDMP TTAC) has compiled the following information on new regulations that have become effective regarding Narcotic Treatment Programs (NTPs), which are now allowed to have mobile sites.
The document informs PDMP administrators, state and federal legislators, and other interested stakeholders about state and federal legislation of each state and federal agencies related to PDMPs. The document identifies bills and regulations introduced at the state and federal levels related to PDMPs.
The Prescription Drug Monitoring Program (PDMP) Model Act 2020 provides state administrators with model language that PDMPs may use when considering developing state laws to ensure that health care practitioners have complete and reliable information regarding their patients’ controlled substance histories and to assist law enforcement in combating drug diversion. The Act includes sections on establishment of advisory boards or committees, reporting, access and use, incorporating alternate data sources, interstate data sharing, audit trail information and transmission audit logs, integration, confidentiality, practitioner activity reports, mandatory registration and query requirements, immunity, data analysis, rules and regulations, unlawful acts, and penalties and severability.
PDMP Introduced and Enacted Legislation and Regulations - Second Quarter 2020
View ArticleArticle — 07/10/2020
The document informs PDMP administrators, state and federal legislators, and other interested stakeholders about state and federal legislation from each state, district, territory, and federal agency related to PDMPs. The document identifies bills and regulations introduced at the state and federal levels related to PDMPs and tracks their progress with quarterly and annual updates and reports.
Prescribing Restrictions for Acute and Chronic Pain - October 2017
View PublicationPublication — 11/27/2017






